Iron-Corroding Bacteria Shown to Possess Enzymes Enabling Them to Extract Electrons from Extracellular Solids Posted on 24 Feb 08:39
—Discovery May Facilitate the Development of Environmental-Friendly Anti-biocorrosion Measures, Such as Enzyme-Targeted Chemicals—
2018.02.14
(2018.02.17 Update)
National Institute for Materials Science (NIMS)
RIKEN
A research team led by NIMS and RIKEN has discovered that bacteria responsible for iron corrosion in petroleum pipelines, etc. possess a group of enzymes enabling them to directly extract electrons from extracellular solids.
Abstract
- A research team led by NIMS and RIKEN has discovered that sulfate-reducing bacteria responsible for anaerobic iron corrosion in petroleum pipelines, etc. possess a group of cell surface enzymes which enable them to directly extract electrons from extracellular solids. Current anticorrosion methods involve the use of antibacterial agents which kill a broad spectrum of bacteria. Their finding may facilitate the development of more efficient and environmental-friendly anti-biocorrosion methods; for example, the formulation of chemicals capable of effectively inhibiting the bacterial enzymes identified in this research.
- Anaerobic iron corrosion in petroleum pipelines cause severe industrial failures, such as oil leakage. It is therefore important to identify the causes of anaerobic corrosion and efficiently prevent them. Sulfate-reducing bacteria—which produce corrosive hydrogen sulfide by oxidizing soluble electron donors such as organics and hydrogen—have been considered as the cause of anaerobic corrosion. However, it remained unknown why corrosion continue proceeding even after iron surfaces were covered with the built-up iron sulfide crusts which protect iron surface from hydrogen sulfide. In 2004, several sulfate-reducing bacteria were isolated with iron as the sole energy source, and hypothesized to be capable of direct electron extraction from iron through electrically conductive of iron sulfide crusts, causing the persistent anaerobic corrosion. However, electron uptake agents such as surface redox enzymes have not been identified in these bacteria, leaving how they extract electrons from solids unknown.
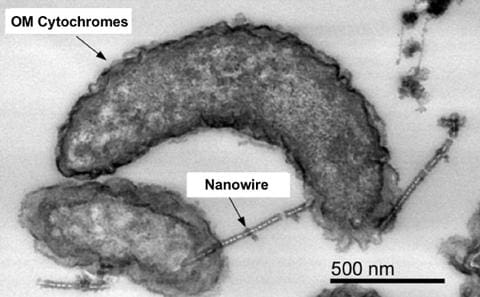
- The research team carefully analyzed the cell membranes of a corrosive sulfate-reducing bacterium which grows with metal iron as the sole electron source, and discovered a group of membrane enzymes (i.e., outer membrane [OM] cytochromes, which are shown in the photo as the dark stains on the cell surface and nanowires). The team confirmed that electrons were removed from an indium-tin doped oxide electrode only when these enzymes were expressed. These results provide strong evidence supporting that this sulfate reducing bacterium can accelerate iron corrosion by direct electron uptake from iron. In addition, the team searched the ubiquity of the newly discovered enzymes in the protein databases and found that the amino acid sequences were widely conserved by various sulfur-metabolizing bacteria inhabiting deep-sea sediments, and distinct from those previously identified in iron-reducing bacteria, therefore likely formed a new clade of outer membrane cytochromes.
- In future studies, the team plans to develop anti-biocorrosion techniques capable of selectively and efficiently deactivating corrosive sulfate-reducing bacteria at low costs in an environmental-friendly manner by designing chemicals which inhibit the electron uptake of the identifed membrane enzymes. The results of this research also indicated the first time that bacteria inhabiting deep-sea sediment—a largely unknown ecosystem—may extract electrons directly from solid matters. These results may facilitate the development of techniques to culture unknown bacteria.
- This research project was carried out by a research team led by Akihiro Okamoto (Senior Researcher, Center for Green Research on Energy and Environmental Materials, NIMS), Xiao Deng (School of Engineering, University of Tokyo; also a recipient of the JSPS Research Fellowship for Young Scientists), Kazuhito Hashimoto (NIMS President and Professor (formally affiliated with the School of Engineering of the University of Tokyo)) and Naoshi Dohmae (Unit Leader, Center for Sustainable Resource Science, RIKEN). This study was conducted as a part of a Specially Promoted Research project (Project No. 24000010) and Young Scientists (A) project (Project No. 17H04969), both funded by Grants-in-Aid for Specially Promoted Research from the Japan Society for the Promotion of Science (JSPS).
- This research was published in Science Advances at 2:00 pm on February 16, 2018, local time (4:00 am on the 17th, Japan Time).